 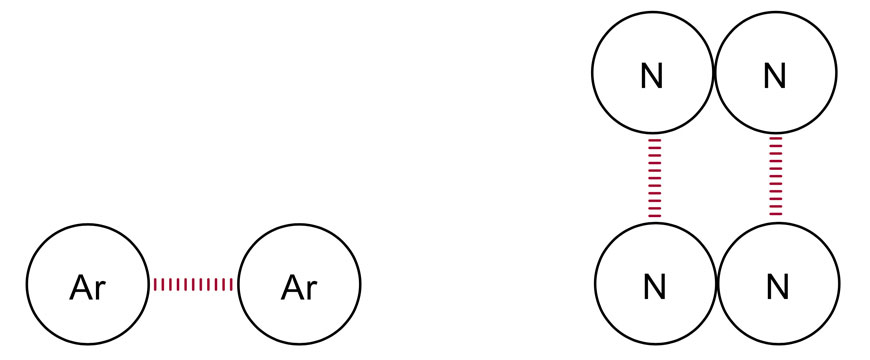
More soluble in polar solvents (E.g water) than non-polar solvents. This intermolecular bond means more energy is required to overcome the potential energy well of the bonds. This means they can form dipole-dipole bonds, a type of intermolecular bond. This is due to the fact that the distribution of charge across the ion isn't uniform, as the bond is polar. However, ionic bonds will always have a dipole, due to the large difference in electronegativity of the two bonding substances. This is due to the fact that for an ionic bond between two ions to occur, the magnitude of the negative charge of one ion must be equal to the magnitude of the positive charge of the other ion. They are electrically neutral, meaning they have a net charge of zero. The formula for an ionic compound is expressed in its simplest ratios. E.g Na 2 Cl 3Ĭoordination number: The number of ions surrounding each ion in the lattice Properties of Ionic Compounds.These consist of ions held together in a lattice structure via electrostatic attraction.Most often between a non-metal and metal. 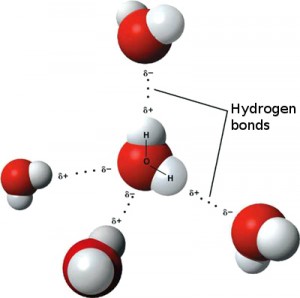
It has a positive charge.Īnion: An ion with more electrons than normally. Cation vs AnionĬation: An ion with fewer electrons than normally. Ions are elements which have had electrons added or removed, giving them either a positive or negative charge. Disclaimer: This only covers the SL information, if you are in HL feel free to edit the page to add the HL content. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
